|
Summary In electrolysis, an external voltage is applied to drive a nonspontaneous reaction. The quantity of material oxidized or reduced can be calculated from the stoichiometry of the reaction and the amount of charge transferred. Relationship of charge, current and time: [ C = A times t nonumber ] In electrolysis, an external voltage is applied to drive a nonspontaneous reaction. Electrolysis can also be used to produce H 2 and O 2 from water. In practice, an additional voltage, called an overvoltage, must be applied to overcome factors such as a large activation energy and a junction potential. Electroplating is the process by which a second metal is deposited on a metal surface, thereby enhancing an object’s appearance or providing protection from corrosion. The amount of material consumed or produced in a reaction can be calculated from the stoichiometry of an electrolysis reaction, the amount of current passed, and the duration of the electrolytic reaction.
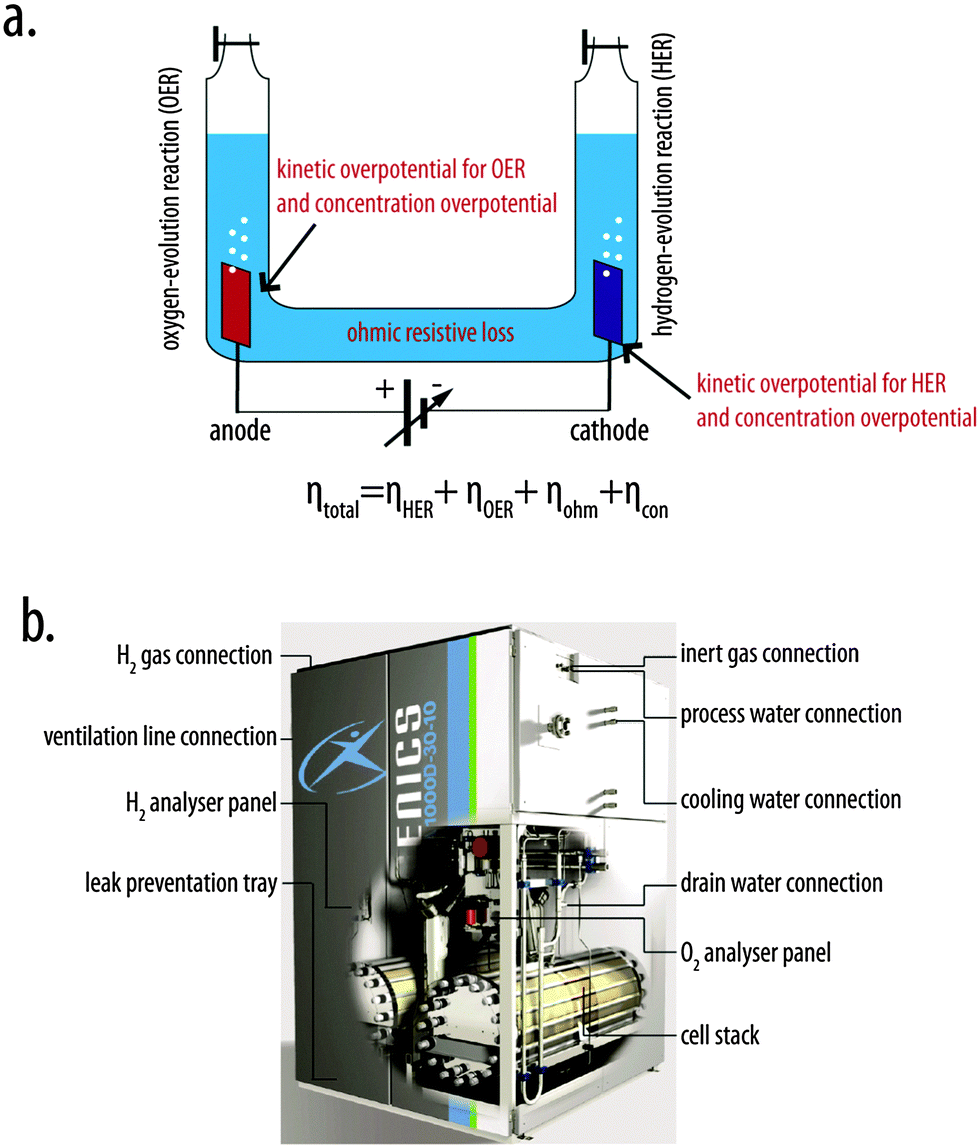
A concentrated solution contains more solute (the chemical dissolved in a solution) particles and fewer water particles than a more dilute acid. - Increasing the concentration of a solution leads to more frequent collisions between reactant particles, so the rate of reaction goes up. METAPHOR - if there were two small rooms, one with 5 people in and the other with 50 people in, the 50 people would have a higher chance of touching each other than the 5 people room because there are more of them. Cycle point of view [6-8]. Powered by solar energy, hydrogen can be produced from water, via photocatalysis, thermochemical cycles, and water electrolysis [9,10]. These methods offer renewable and clean production of hydrogen fuel and, therefore, have attracted increasing research interests in recent years. Hydrogen production via electrolysis of water (water-splitting reaction) is a means of storing excess electrical energy produced by renewable energy sources. This hydrogen gas may be used directly to produce power via combustion or recombination with oxygen in a fuel cell, it may be injected into the natural gas network, and it may be used as a. PHYSICAL SETTING CHEMISTRY Wednesday, June 16, 2010 — 1.15 to 4.15 p.m., only. Activation energy (2) entropy of the system (3) heat of fusion (4) heat of reaction. Electrolysis of 10. G of water (3) nuclear fission of 10. G of uranium (4) oxidation of 10. So, the activation energy expresses the speed with which a reaction takes place. The balance energy, per mole, during water electrolysis is shown in Fig.2. The design of the second PEM water electrolysis demonstration plant, Solar. An experiment was designed to investigate the water electrolysis process. Act,i the activation energy a, c anode, cathode A surface area, cm2 N: H2 hydrogen producing rate, mol s 1 N: O2 oxygen producing rates, mol s 1. A concise model for evaluating water electrolysis. To increase the rate of a reaction you need to increase the number of successful collisions. One possible way of doing this is to provide an alternative way for the reaction to happen which has a lower activation energy. Adding a catalyst has exactly this effect on activation energy. A catalyst provides an alternative route for the reaction. That alternative route has a lower activation energy. Be very careful if you are asked about this in an exam. The correct form of words is Computer link software ti nspire cx. 'A catalyst provides an alternative route for the reaction with a lower activation energy.' Energy Required For Electrolysis32bit web browser 11 01 26 executive park. It does not 'lower the activation energy of the reaction'. There is a subtle difference between the two statements that is easily illustrated with a simple analogy. Activation Energy Of Water Electrolysis DemonstrationsTEMPERATURE * See diagram* Sodium thiosulfate and hydrochloric acid react to make a sulphur (a solid) which makes the solution go cloudy. You can measure the rate of reaction by timing how long it takes for the solution to become cloudy. Sodium thiosulfate + hydrochloric acid --> sodium chloride + sulphur dioxide + sulphur + water We repeated this experiment with the conical flask containing the solution in different temperatures. Electrolysis Of WaterThe reaction occurred fastest under the highest temperature - the cross disappeared the fastest. HIGHER TEMPERATURE = FASTER REACTION.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
